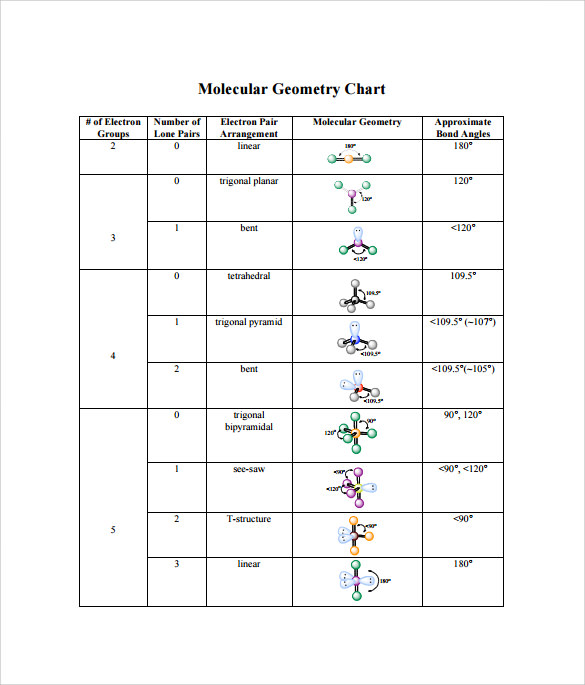
The Geometry of the Molecules of Be, B, and CĨ.5.
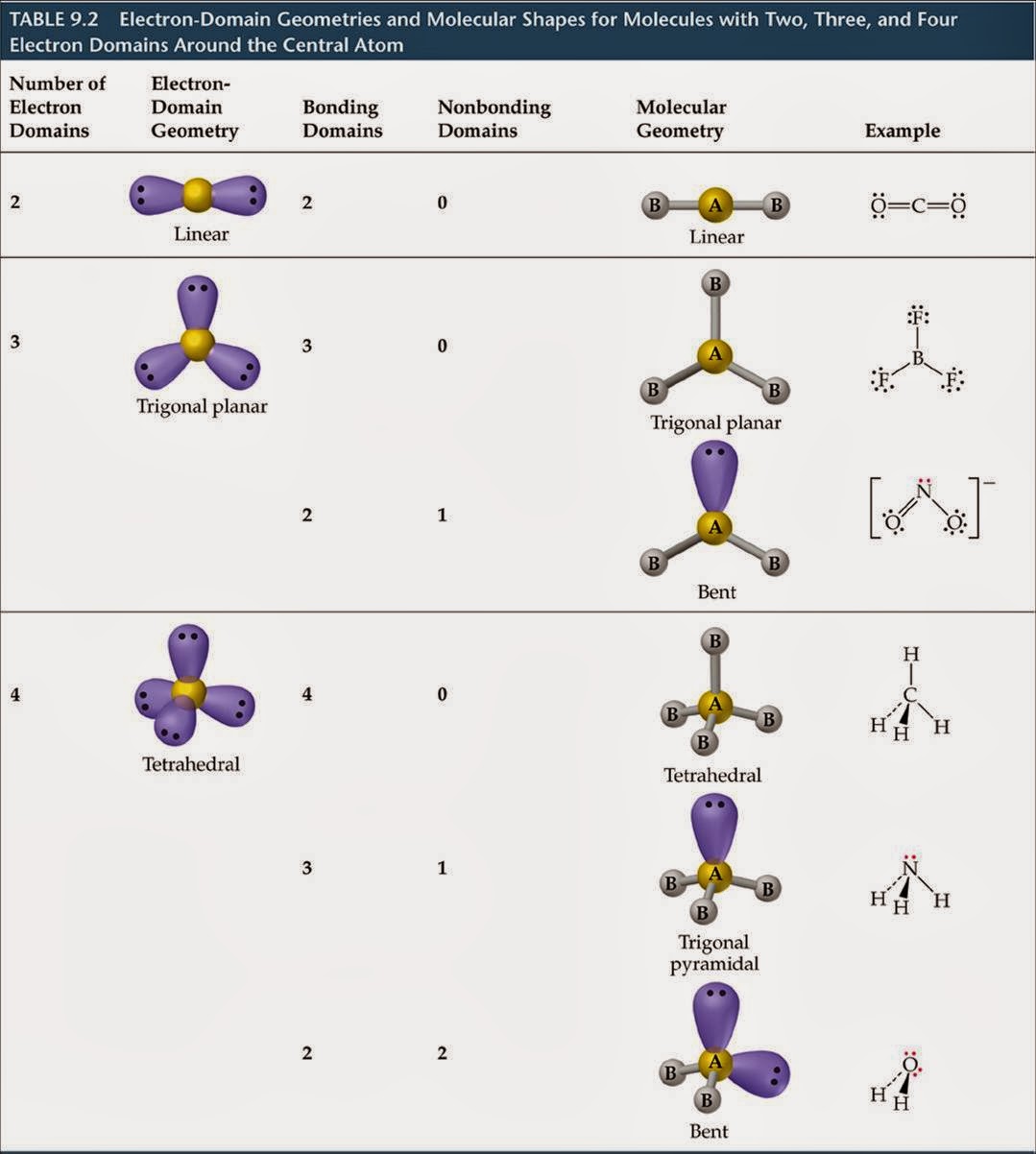
The Nature of the Bonding in the Fluorides, Chlorides, and Hydrides of Li, Be, B, and CĨ.4. The Relationship between Bond Properties and the AIM TheoryĨ.3. Electron Pair Localization and the Lewis and VSEPR ModelsĬhapter 8 Molecules of the Elements of Period 2Ĩ.2. The Valence Shell Charge Concentrationħ.5.
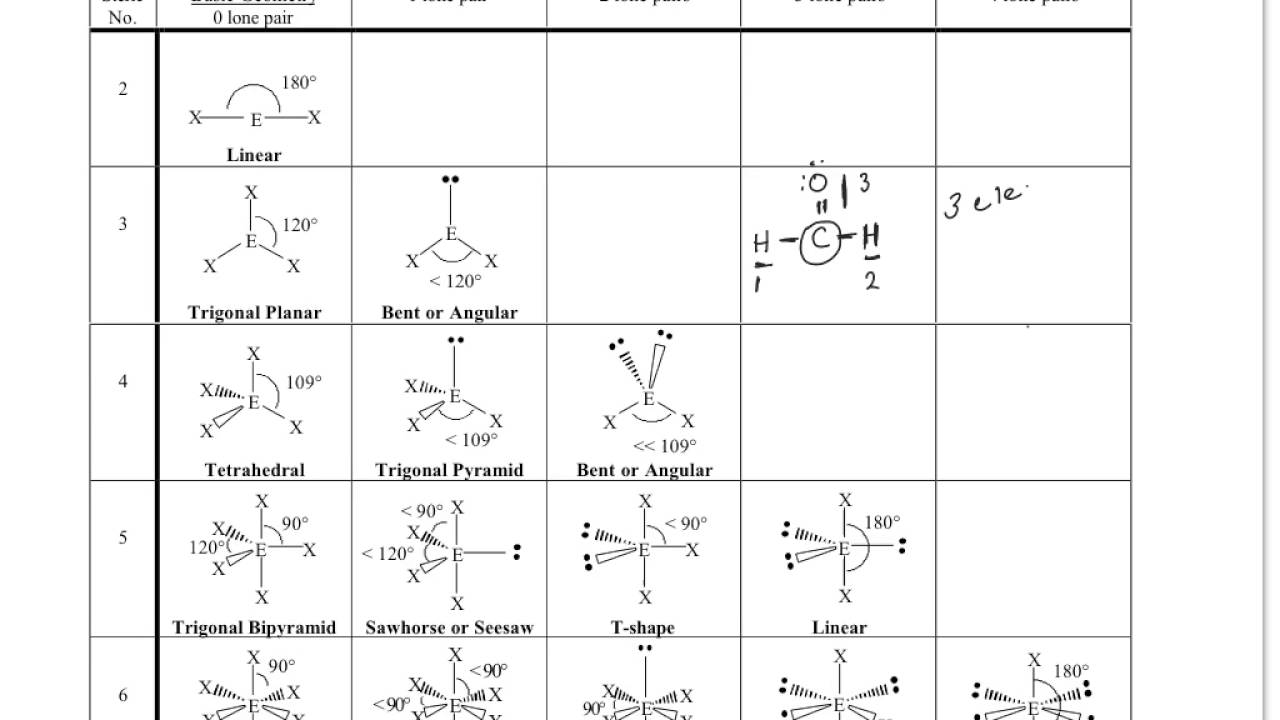
The Laplacian of the Electron Densityħ.3. The Diatomic Hydrides of Periods 2 and 3Ĭhapter 7 The Laplacian of the Electron Densityħ.2. The Density Difference or Deformation FunctionĦ.5. Comparison of the LCP and VSEPR ModelsĬhapter 6 The AIM Theory and the analysis of the electron densityĦ.4. Ligand-Ligand Interactions in Molecules of the Elements in Periods 3-6ĥ.10. Bond Angles in Molecules with Lone Pairsĥ.8. Molecules with Two or More Different Ligandsĥ.6. Two, Three, Four, and Six Electron Pair Valence ShellsĬhapter 5 Ligand-Ligand Interactions and the Ligand Close-Packing (LCP) Modelĥ.3. The Distribution of Electrons in Valence ShellsĤ.4. Multielectron Atoms and Electron ConfigurationsĬhapter 4 Molecular Geometry and the VSEPR ModelĤ.2. The Hydrogen Atom and Atomic Orbitalsģ.10. The Meaning of the Wave Function: Probability and Electron Densityģ.7. The Schrödinger Equation and the Wave Functionģ.6. The Wave Nature of Matter and the Uncertainty Principleģ.5. Bond Dissociation Energies and Bond EnthalpiesĬhapter 3 Some Basic Concepts of Quantum Mechanicsģ.2. Exceptions to the Octet Rule: Hypervalent and Hypovalent MoleculesĢ.7. Polyatomic Anions and Formal Chargesġ.13. The Covalent Bond and Lewis Structuresġ.10. Of electron density distributions using the atoms in molecules (AIM) theory * Emphasizes the relationships between these various models, giving examples of their uses, limitations, and comparative advantages and disadvantagesĬhapter 1 The Chemical Bond: Classical Concepts and Theoriesġ.7. Its earliest days, through Lewis's brilliant concept of the electron pair bond and up to the present day * Presents a novel, non-traditional approach that emphasizes the importance of the Pauli principle as a basis for understanding bonding * Begins with the fundamental classical concepts and proceeds through orbital models to recent ideas based on the analysis of electron densities, which help to clarify and emphasize many of the limitations of earlier models * Provides a thorough and up-to-date treatment of the well-known valence-shell electron pair (VSEPR) model (which was first formulated and developed by author Ronald Gillespie) and the more recent ligand close-packing (LCP) model * Presents a unique pictorial and nonmathematical discussion of the analysis Features: * Shows students how the concept of the chemical bond has developed from

It also serves as a useful resource and an up-to-date introduction to modern developments in the field for instructors teaching chemical bonding at any level. Authored by Ronald Gillespie, a world-class chemist and expert on chemical bonding, and Paul Popelier of the University of Manchester Institute of Science and Technology, this text provides students with a comprehensive and detailed introduction to the principal models and theories of chemical bonding and geometry. Ideal for undergraduate and first-year graduate courses in chemical bonding, Chemical Bonding and Molecular Geometry: From Lewis to Electron Densities can also be used in inorganic chemistry courses.
